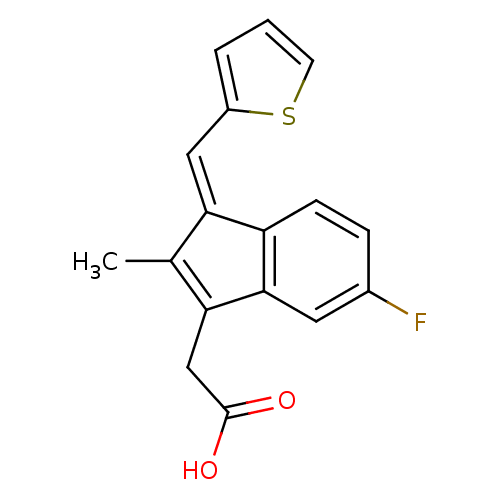
 BDBM84515 Sulindac analogue, 12
BDBM84515 Sulindac analogue, 12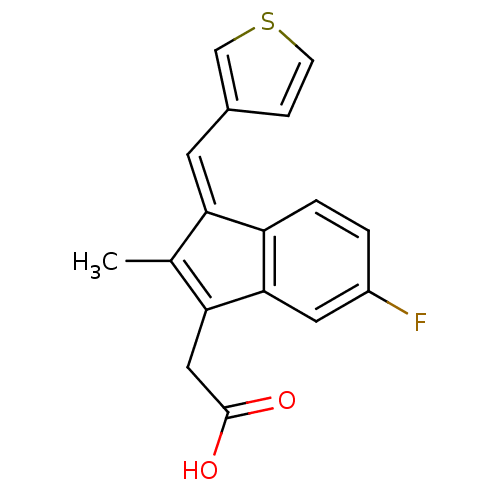 BDBM84516 Sulindac analogue, 13
BDBM84516 Sulindac analogue, 13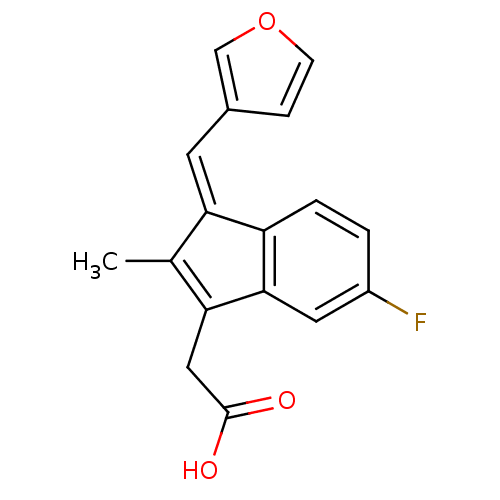 BDBM84517 Sulindac analogue, 14
BDBM84517 Sulindac analogue, 14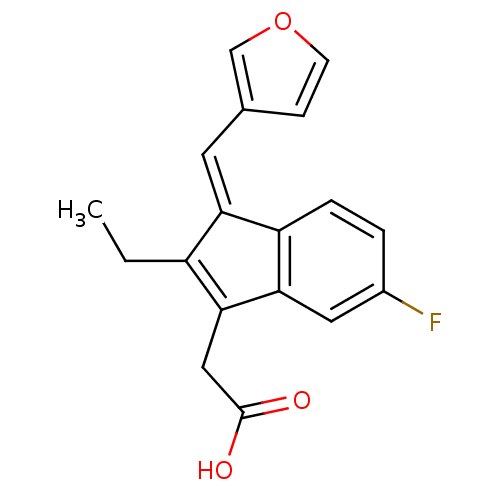 BDBM84518 Sulindac analogue, 15
BDBM84518 Sulindac analogue, 15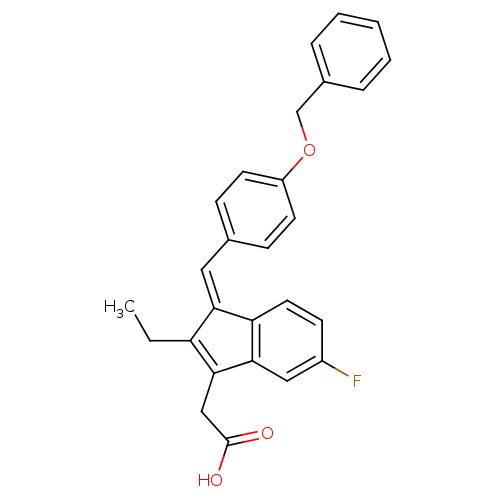 BDBM84519 Sulindac analogue, 16
BDBM84519 Sulindac analogue, 16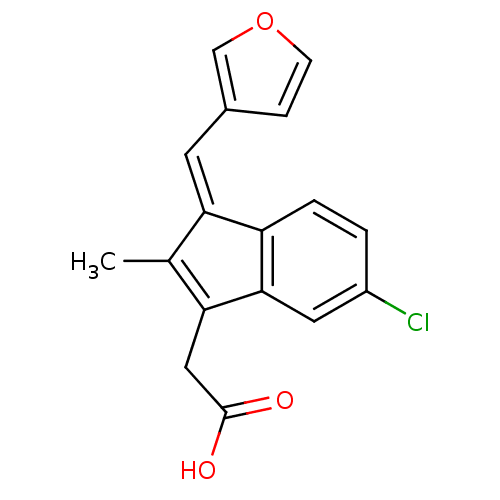 BDBM84520 Sulindac analogue, 17
BDBM84520 Sulindac analogue, 17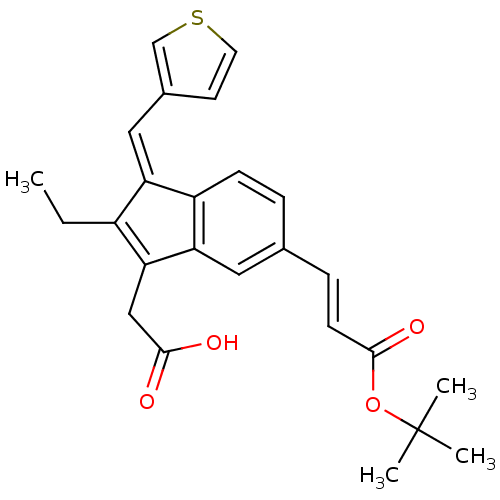 BDBM84526 Sulindac analogue, 23
BDBM84526 Sulindac analogue, 23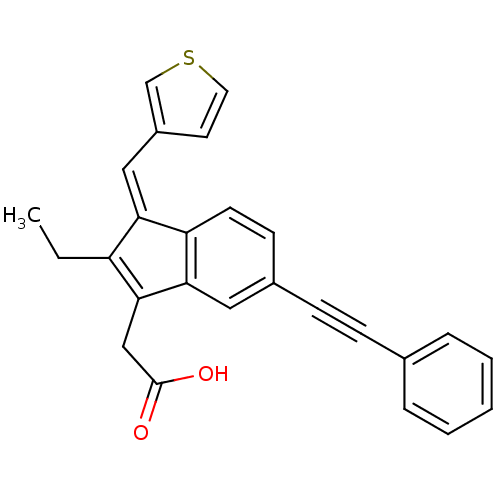 BDBM84527 Sulindac analogue, 24
BDBM84527 Sulindac analogue, 24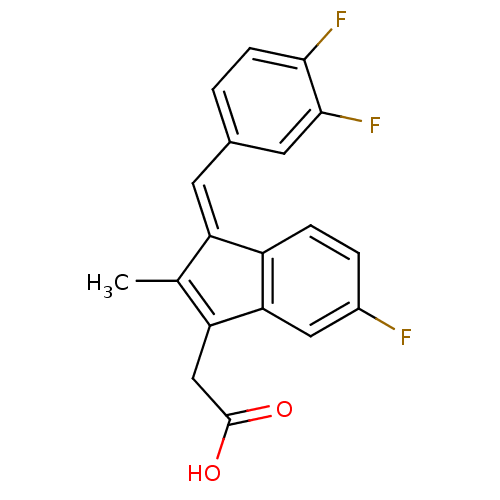 Sulindac analogue, 10 BDBM84513
Sulindac analogue, 10 BDBM84513 Sulindac analogue, 11 BDBM84514
Sulindac analogue, 11 BDBM84514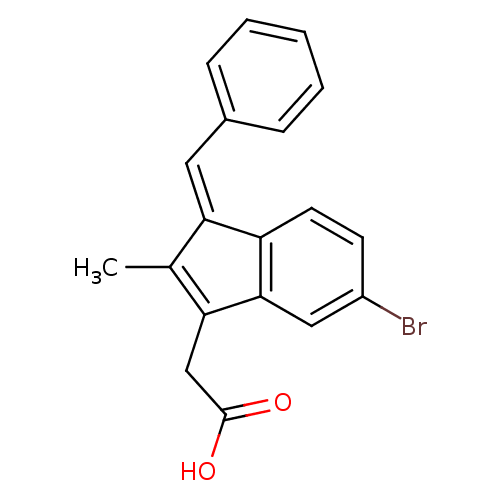 Sulindac analogue, 18 BDBM84521
Sulindac analogue, 18 BDBM84521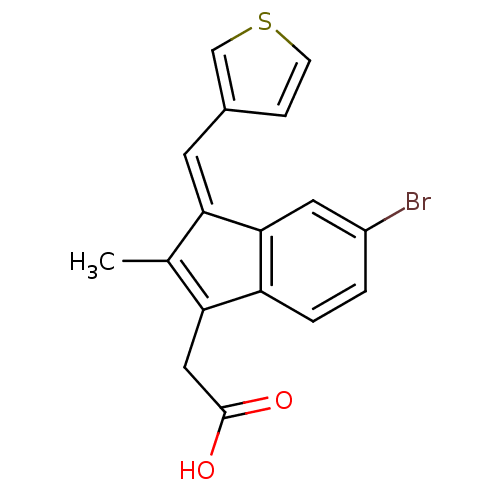 Sulindac analogue, 19 BDBM84522
Sulindac analogue, 19 BDBM84522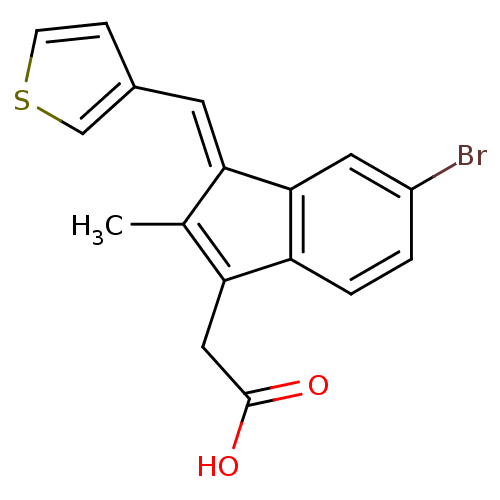 Sulindac analogue, 20 BDBM84523
Sulindac analogue, 20 BDBM84523 Sulindac analogue, 21 BDBM84524
Sulindac analogue, 21 BDBM84524 Sulindac analogue, 22 BDBM84525
Sulindac analogue, 22 BDBM84525 Sulindac Clinoril CHEBI:9352 BDBM50103504
Sulindac Clinoril CHEBI:9352 BDBM50103504 BDBM50293599 (Z)-2'-des-methyl sulindac sulfide CHEMBL561959
BDBM50293599 (Z)-2'-des-methyl sulindac sulfide CHEMBL561959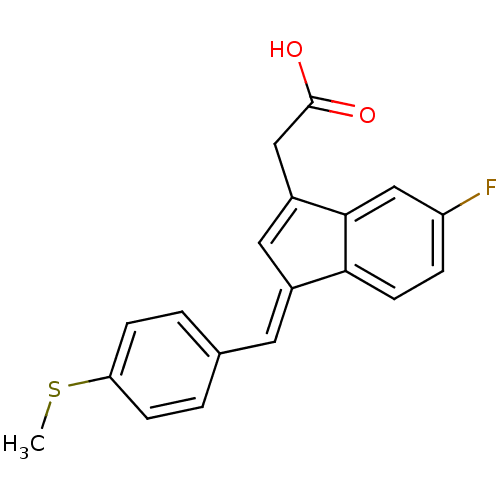 BDBM50272862 (E)-2'-des-methyl sulindac sulfide 2-desmethylsulindac sulfide CHEMBL496464
BDBM50272862 (E)-2'-des-methyl sulindac sulfide 2-desmethylsulindac sulfide CHEMBL496464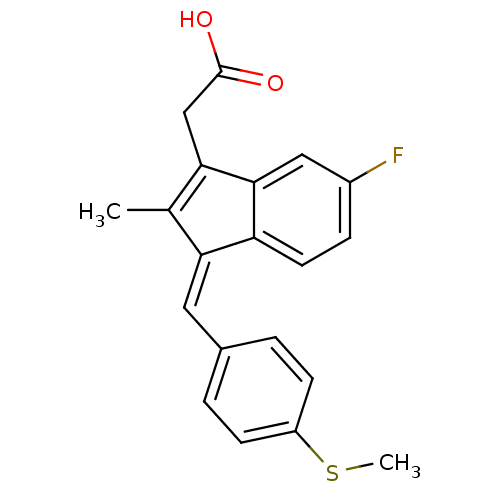 CHEMBL18797 [6-Fluoro-2-methyl-3-(4-methylsulfanyl-benzylidene)-3H-inden-1-yl]-acetic acid cid_5352624 BDBM50110164 2-(3-(4-(methylthio)benzylidene)-6-fluoro-2-methyl-3H-inden-1-yl)acetic acid SULINDAC SULFIDE {6-Fluoro-2-methyl-3-[1-(4-methylsulfanyl-phenyl)-meth-(E)-ylidene]-3H-inden-1-yl}-acetic acid {6-Fluoro-2-methyl-3-[1-[4-(methyl-lambda*4*-sulfanyl)-phenyl]-meth-(E)-ylidene]-3H-inden-1-yl}-acetic acid Sulindac sulphide (Z)-2-(3-(4-(methylthio)benzylidene)-6-fluoro-2-methyl-3H-inden-1-yl)acetic acid
CHEMBL18797 [6-Fluoro-2-methyl-3-(4-methylsulfanyl-benzylidene)-3H-inden-1-yl]-acetic acid cid_5352624 BDBM50110164 2-(3-(4-(methylthio)benzylidene)-6-fluoro-2-methyl-3H-inden-1-yl)acetic acid SULINDAC SULFIDE {6-Fluoro-2-methyl-3-[1-(4-methylsulfanyl-phenyl)-meth-(E)-ylidene]-3H-inden-1-yl}-acetic acid {6-Fluoro-2-methyl-3-[1-[4-(methyl-lambda*4*-sulfanyl)-phenyl]-meth-(E)-ylidene]-3H-inden-1-yl}-acetic acid Sulindac sulphide (Z)-2-(3-(4-(methylthio)benzylidene)-6-fluoro-2-methyl-3H-inden-1-yl)acetic acid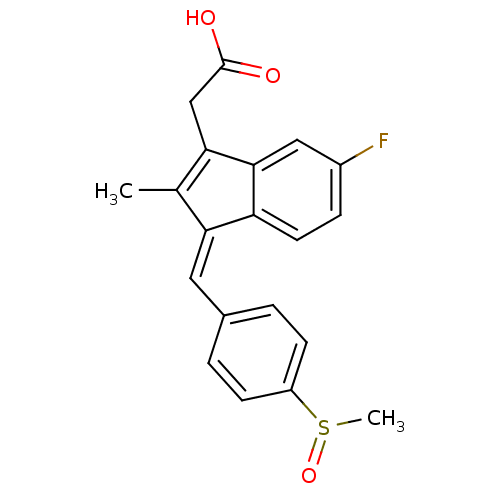 (Z)-5-Fluoro-2-methyl-1-((p-(methylsulfinyl)phenyl)methylene)-1H-indene-3-acetic acid BDBM50012899 CHEMBL15770 cis-5-Fluoro-2-methyl-1-((4-(methylsulfinyl)phenyl)methylene)-1H-indene-3-acetic acid {(1Z)-5-fluoro-2-methyl-1-[4-(methylsulfinyl)benzylidene]-1H-inden-3-yl}acetic acid cis-5-Fluoro-2-methyl-1-((p-methylsulfinyl)benzylidene)indene-3-acetic acid SULINDAC
(Z)-5-Fluoro-2-methyl-1-((p-(methylsulfinyl)phenyl)methylene)-1H-indene-3-acetic acid BDBM50012899 CHEMBL15770 cis-5-Fluoro-2-methyl-1-((4-(methylsulfinyl)phenyl)methylene)-1H-indene-3-acetic acid {(1Z)-5-fluoro-2-methyl-1-[4-(methylsulfinyl)benzylidene]-1H-inden-3-yl}acetic acid cis-5-Fluoro-2-methyl-1-((p-methylsulfinyl)benzylidene)indene-3-acetic acid SULINDAC
- Gourzoulidou, E; Carpintero, M; Baumhof, P; Giannis, A; Waldmann, H Inhibition of angiogenesis-relevant receptor tyrosine kinases by sulindac analogues. Chembiochem 6: 527-31 (2005)
- Walters, MJ; Blobaum, AL; Kingsley, PJ; Felts, AS; Sulikowski, GA; Marnett, LJ The influence of double bond geometry in the inhibition of cyclooxygenases by sulindac derivatives. Bioorg Med Chem Lett 19: 3271-4 (2009)
- Felts, AS; Siegel, BS; Young, SM; Moth, CW; Lybrand, TP; Dannenberg, AJ; Marnett, LJ; Subbaramaiah, K Sulindac derivatives that activate the peroxisome proliferator-activated receptor gamma but lack cyclooxygenase inhibition. J Med Chem 51: 4911-9 (2008)
- Liedtke, AJ; Crews, BC; Daniel, CM; Blobaum, AL; Kingsley, PJ; Ghebreselasie, K; Marnett, LJ Cyclooxygenase-1-selective inhibitors based on the (E)-2'-des-methyl-sulindac sulfide scaffold. J Med Chem 55: 2287-300 (2012)
- Huang, F; Zeng, Z; Zhang, W; Yan, Z; Chen, J; Yu, L; Yang, Q; Li, Y; Yu, H; Chen, J; Wu, C; Zhang, XK; Su, Y; Zhou, H Design, synthesis, and biological evaluation of novel sulindac derivatives as partial agonists of PPAR? with potential anti-diabetic efficacy. Eur J Med Chem 222: (2021)
 BDBM84515 Sulindac analogue, 12
BDBM84515 Sulindac analogue, 12 BDBM84516 Sulindac analogue, 13
BDBM84516 Sulindac analogue, 13 BDBM84517 Sulindac analogue, 14
BDBM84517 Sulindac analogue, 14 BDBM84518 Sulindac analogue, 15
BDBM84518 Sulindac analogue, 15 BDBM84519 Sulindac analogue, 16
BDBM84519 Sulindac analogue, 16 BDBM84520 Sulindac analogue, 17
BDBM84520 Sulindac analogue, 17 BDBM84526 Sulindac analogue, 23
BDBM84526 Sulindac analogue, 23 BDBM84527 Sulindac analogue, 24
BDBM84527 Sulindac analogue, 24 Sulindac analogue, 10 BDBM84513
Sulindac analogue, 10 BDBM84513 Sulindac analogue, 11 BDBM84514
Sulindac analogue, 11 BDBM84514 Sulindac analogue, 18 BDBM84521
Sulindac analogue, 18 BDBM84521 Sulindac analogue, 19 BDBM84522
Sulindac analogue, 19 BDBM84522 Sulindac analogue, 20 BDBM84523
Sulindac analogue, 20 BDBM84523 Sulindac analogue, 21 BDBM84524
Sulindac analogue, 21 BDBM84524 Sulindac analogue, 22 BDBM84525
Sulindac analogue, 22 BDBM84525 Sulindac Clinoril CHEBI:9352 BDBM50103504
Sulindac Clinoril CHEBI:9352 BDBM50103504 BDBM50293599 (Z)-2'-des-methyl sulindac sulfide CHEMBL561959
BDBM50293599 (Z)-2'-des-methyl sulindac sulfide CHEMBL561959 BDBM50272862 (E)-2'-des-methyl sulindac sulfide 2-desmethylsulindac sulfide CHEMBL496464
BDBM50272862 (E)-2'-des-methyl sulindac sulfide 2-desmethylsulindac sulfide CHEMBL496464 CHEMBL18797 [6-Fluoro-2-methyl-3-(4-methylsulfanyl-benzylidene)-3H-inden-1-yl]-acetic acid cid_5352624 BDBM50110164 2-(3-(4-(methylthio)benzylidene)-6-fluoro-2-methyl-3H-inden-1-yl)acetic acid SULINDAC SULFIDE {6-Fluoro-2-methyl-3-[1-(4-methylsulfanyl-phenyl)-meth-(E)-ylidene]-3H-inden-1-yl}-acetic acid {6-Fluoro-2-methyl-3-[1-[4-(methyl-lambda*4*-sulfanyl)-phenyl]-meth-(E)-ylidene]-3H-inden-1-yl}-acetic acid Sulindac sulphide (Z)-2-(3-(4-(methylthio)benzylidene)-6-fluoro-2-methyl-3H-inden-1-yl)acetic acid
CHEMBL18797 [6-Fluoro-2-methyl-3-(4-methylsulfanyl-benzylidene)-3H-inden-1-yl]-acetic acid cid_5352624 BDBM50110164 2-(3-(4-(methylthio)benzylidene)-6-fluoro-2-methyl-3H-inden-1-yl)acetic acid SULINDAC SULFIDE {6-Fluoro-2-methyl-3-[1-(4-methylsulfanyl-phenyl)-meth-(E)-ylidene]-3H-inden-1-yl}-acetic acid {6-Fluoro-2-methyl-3-[1-[4-(methyl-lambda*4*-sulfanyl)-phenyl]-meth-(E)-ylidene]-3H-inden-1-yl}-acetic acid Sulindac sulphide (Z)-2-(3-(4-(methylthio)benzylidene)-6-fluoro-2-methyl-3H-inden-1-yl)acetic acid (Z)-5-Fluoro-2-methyl-1-((p-(methylsulfinyl)phenyl)methylene)-1H-indene-3-acetic acid BDBM50012899 CHEMBL15770 cis-5-Fluoro-2-methyl-1-((4-(methylsulfinyl)phenyl)methylene)-1H-indene-3-acetic acid {(1Z)-5-fluoro-2-methyl-1-[4-(methylsulfinyl)benzylidene]-1H-inden-3-yl}acetic acid cis-5-Fluoro-2-methyl-1-((p-methylsulfinyl)benzylidene)indene-3-acetic acid SULINDAC
(Z)-5-Fluoro-2-methyl-1-((p-(methylsulfinyl)phenyl)methylene)-1H-indene-3-acetic acid BDBM50012899 CHEMBL15770 cis-5-Fluoro-2-methyl-1-((4-(methylsulfinyl)phenyl)methylene)-1H-indene-3-acetic acid {(1Z)-5-fluoro-2-methyl-1-[4-(methylsulfinyl)benzylidene]-1H-inden-3-yl}acetic acid cis-5-Fluoro-2-methyl-1-((p-methylsulfinyl)benzylidene)indene-3-acetic acid SULINDAC